28 Jun The 9-valent HPV vaccine is now available for free! (Since April 2023)
Steps you can take now to reduce your risk of getting cervical cancer.
What is the HPV vaccine?
The HPV vaccine protects against infection with the human papillomavirus, the main cause of cervical cancer. Apart from the 9 valent vaccine (HPV9), there is a bivalent vaccine (HPV2) and quadrivalent vaccine (HPV4). Please consult with your doctor about which vaccine you should use.
What is the 9-valent HPV vaccine?
There are many types of HPV. The 9-valent vaccine prevents infection with nine of these HPV types. Seven* of these types are responsible for 80% to 90% of cervical cancers.
*Types 16, 18, 31, 33, 45, 52 and 58
Are there any side effects with the 9-valent vaccine?
You may have some of the side effects listed in the following table after receiving the 9-valent vaccine.
If you experience any physical changes or any concerning symptoms after vaccination, you should initially consult a doctor at the clinic/hospital where you received the vaccine.
Symptoms that may occur following vaccination.
| Frequency of Occurrence | Reported Symptoms |
| 50% or more | *Pain |
| 10% to less than 50% | Swelling, erythema (redness), headache |
| 1% to less than 10% |
Dizziness (lightheadedness), nausea, diarrhea, *itching, fever, fatigue, *bruising, etc. |
| Less than 1% |
Vomiting, abdominal pain, sore muscles, joint pain, bleeding*, hematoma*, malaise (lethargy), hardening of the skin, etc. |
| Frequency unknown |
Hypoesthesia (decreased sensitivity to stimuli), fainting, pain in the extremities, etc. |
*Symptoms at the site of vaccination
Adapted from the attached Silgard®9 document (1st edition)
This cancer could affect YOU!
- Cervical cancer is a cancer that develops at the entrance to the uterus, called the cervix.In Japan, approximately 11,000 women are diagnosed with cervical cancer every year and about 2,900 women die from it.
- The frequency of cervical cancer is relatively high in younger women. It starts to increase in women in their 20s and around 1,000 women each year have to have their uterus removed by the age of 40 due to treatment for cervical cancer. In Japan, cervical cancer is the second leading cause of cancer death among women aged 25 to 40.
- While the HPV vaccine is expected to protect against cervical cancer by preventing infection with HPV, some HPV infections cannot be prevented by the vaccine. In order to be able to detect and treat the cancer early, it is important for women to attend cervical screening once every two years from the age of 20.
Who is eligible?
Girls in the sixth grade of elementary school to the first year of high school. or equivalent ages (age 12 to 16). are eligible for the vaccination.
Those eligible for the catch-up vaccination program in 2023: Women born between April 2, 1997 and April 1, 2006
Catch-up vaccination period: April 1, 2022 to March 31, 2025
For further information please refer to the 9-valent HPV Vaccination Leaflet (Catch-up Program)(Japanese)
General Vaccination Schedule
Those who start vaccination with the 9-valent vaccine (Silgard®9) will receive a total of 2 or 3 doses at set intervals. If you experience any concerning symptoms after the first or second dose, you can choose not to receive any further doses.
To those who have already received one or two doses of the bivalent or quadrivalent HPV vaccine (Cervarix® or Gardasil®)
In general, we recommend that you receive the same type of vaccine as your previous doses. In consultation with your doctor, however, you can switch to the 9-valent vaccine during the course of vaccination to complete the remaining doses. In this case, you can still receive the vaccination at public expense.
For further information please refer to:
The Cervical Cancer Vaccine (2vHPV and 4vHPV vaccine) in Sapporo as of April 2022
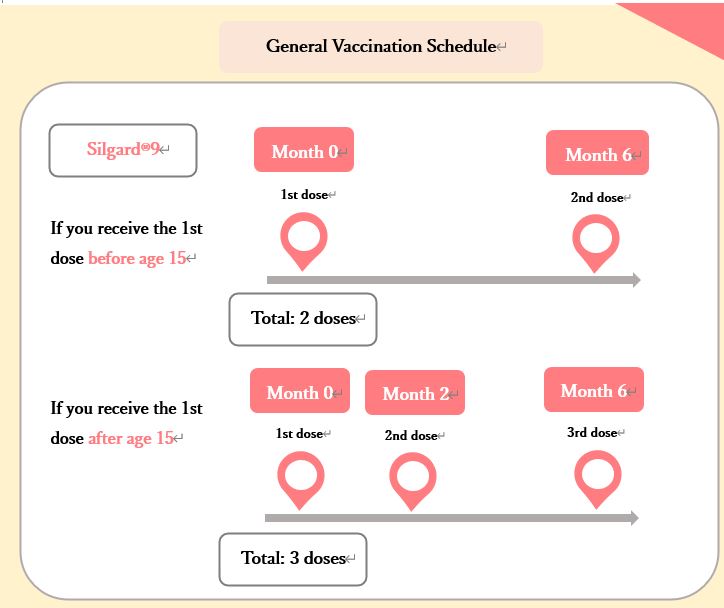
In either case, it is desirable that all doses are completed within one year.
Based on the information from the 9-valent HPV Vaccination Leaflet (Routine Vaccination Program) by Ministry of Health, Labour and Welfare


